Researchers at Indian Institute of Science made a breakthrough discovery Tech Explore says could replace conventional lithium-ion batteries. They published their paper Reactive Electrolyte Additives Improve Lithium Metal Battery Performance on December 17, 2021, although news is only reaching main stream media now. Essentially, they found electrolyte additives increase performance of solid state lithium batteries without dendrites forming.
Enhanced Lithium Batteries Without Dendrites in Solid State
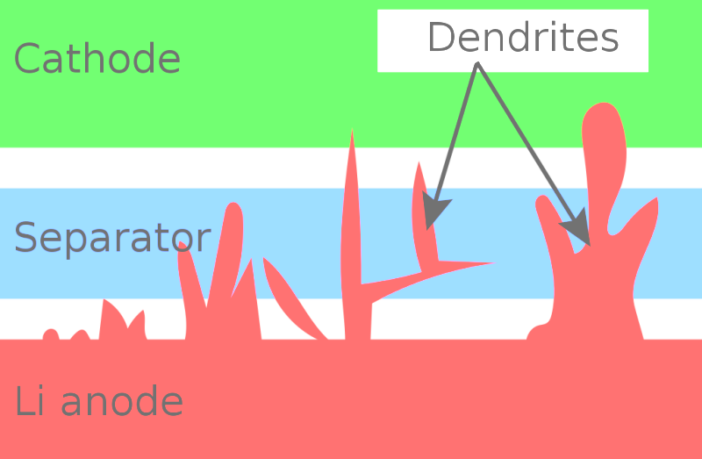
Solid state lithium batteries have made considerable inroads in portable electronics. However, eventually dendrites form causing electrodes to short circuit and destroy the cell. But the Indian Institute of Science researchers say they now know why this happens.
That’s because their study reveals microscopic voids form in one of the electrodes early on in battery life. However, they are able to delay dendrites forming by plating the solid electrolyte surface with “a thin layer of certain metals”. This in turn extends the battery’s life, and enables faster charging.
“This work presents a breakthrough in the development of electrolyte additives for high-energy-density li-ion batteries,” the team explains. “We expect our systematic approach for rational molecular design, and density function theory-aided development offers a promising way to discover next-generation additives.”
How the Team Reached This Startling Simple Solution
The team from Indian Institute of Science followed a logical scientific approach to reach their discovery:
- First, they artificially induced dendrite formation while studying micro thin battery slices under a scanning electron microscope.
- As they did do, they realized microscopic voids were developing in the lithium anode during discharge.
- Then they discovered the currents concentrated at the edges of these microscopic voids were about 10,000 times larger than usual.
- This in turn led them to the conclusion this was likely creating stress on the solid electrolyte, and accelerating the dendrite formation.
Their solution was simple. Introduce an ultra-thin layer of a metal that is resistant to heat and wear, between the lithium anode and solid electrolyte separator. This refractory metal layer shields the solid electrolyte from the stress, and redistributes the current to an extent they confirm.
Breaking News
Faster Plating With Sodium Glassy Electrodes
Is Power to Gas Storage The Next Battery